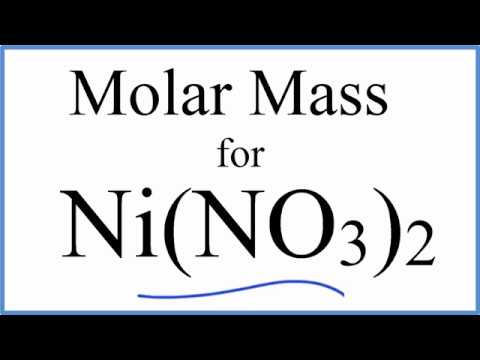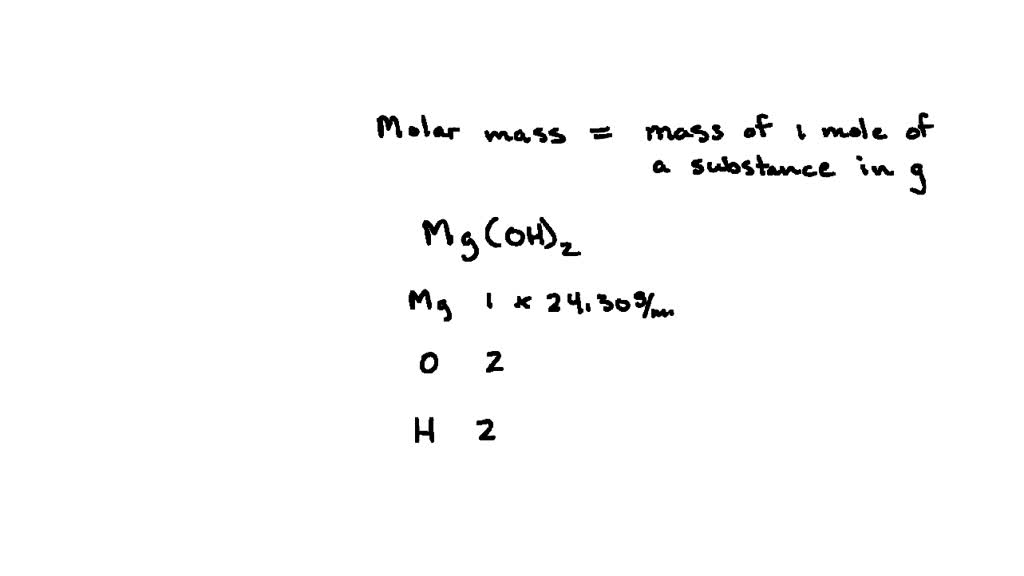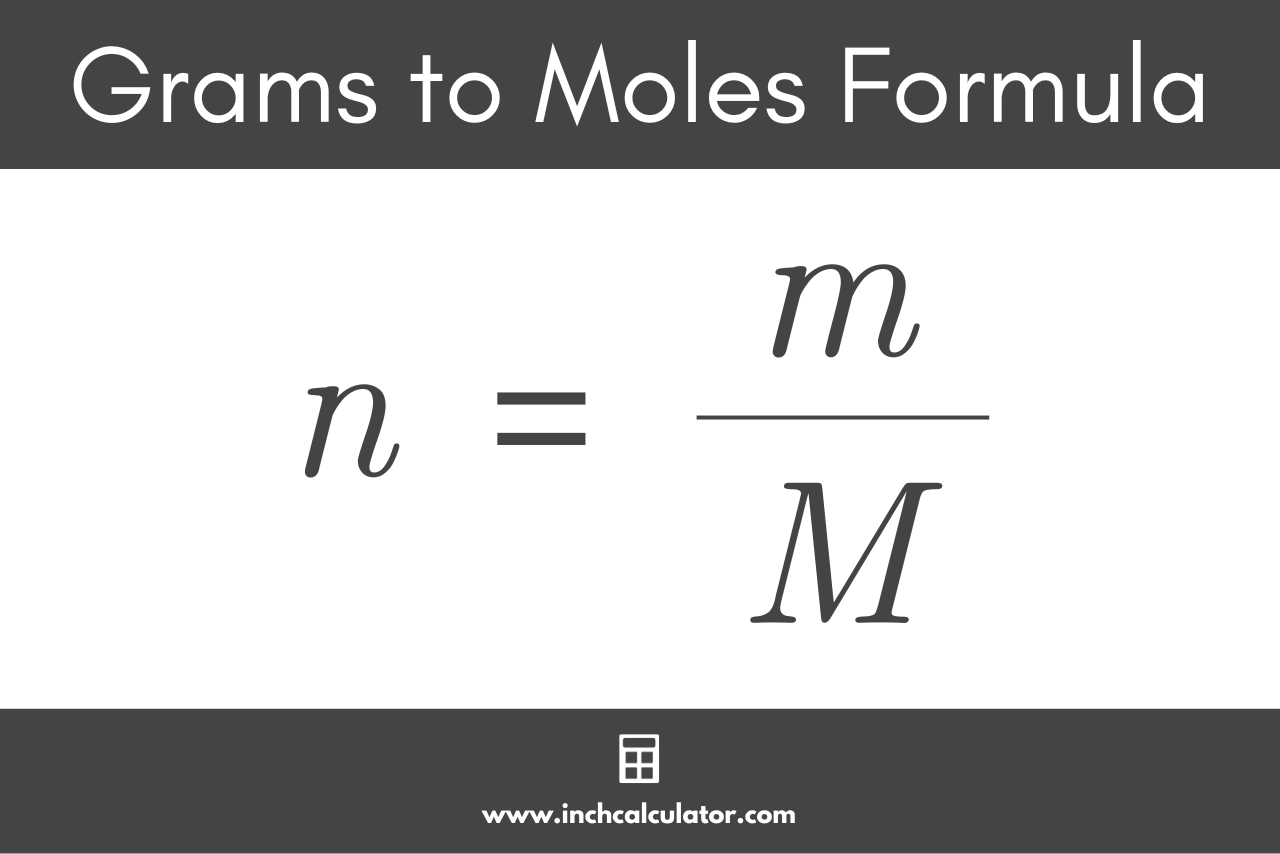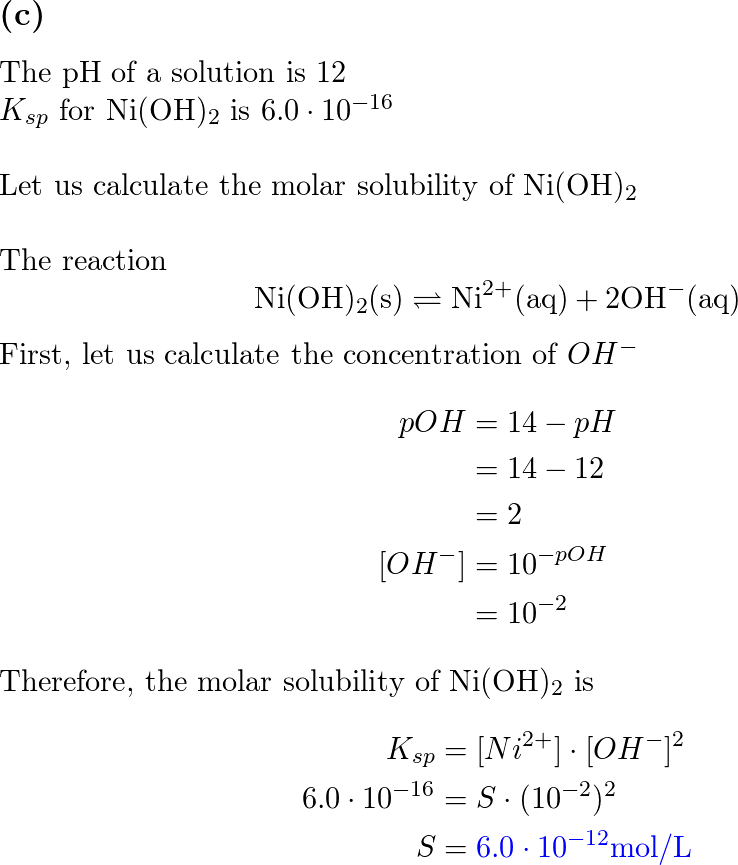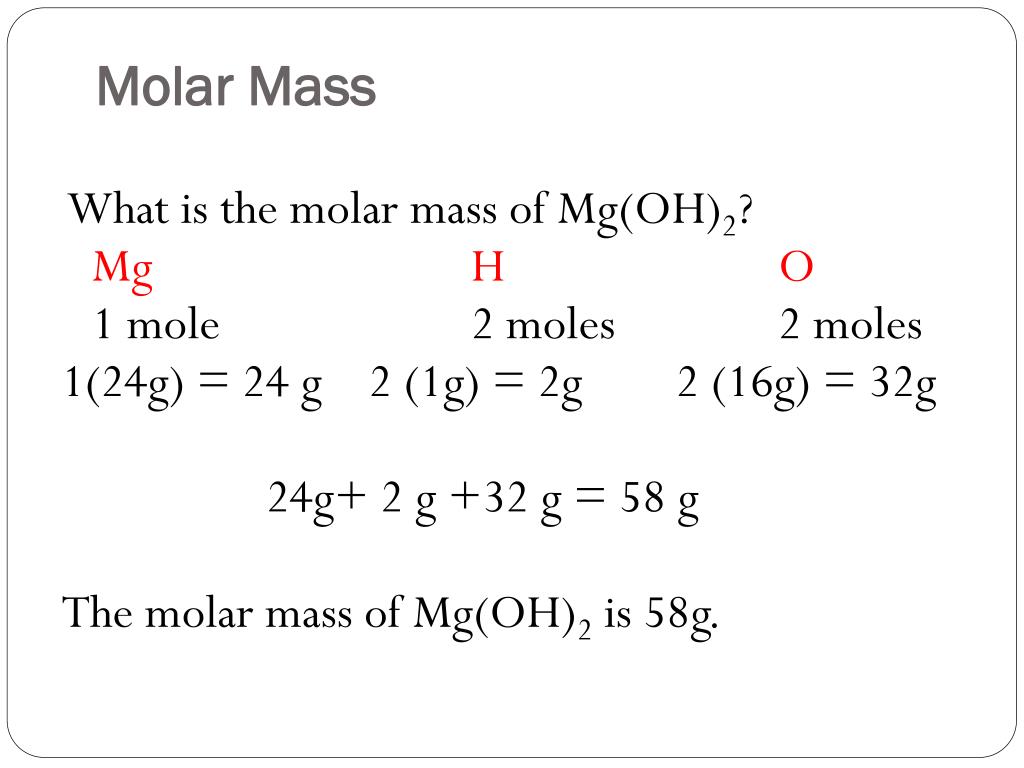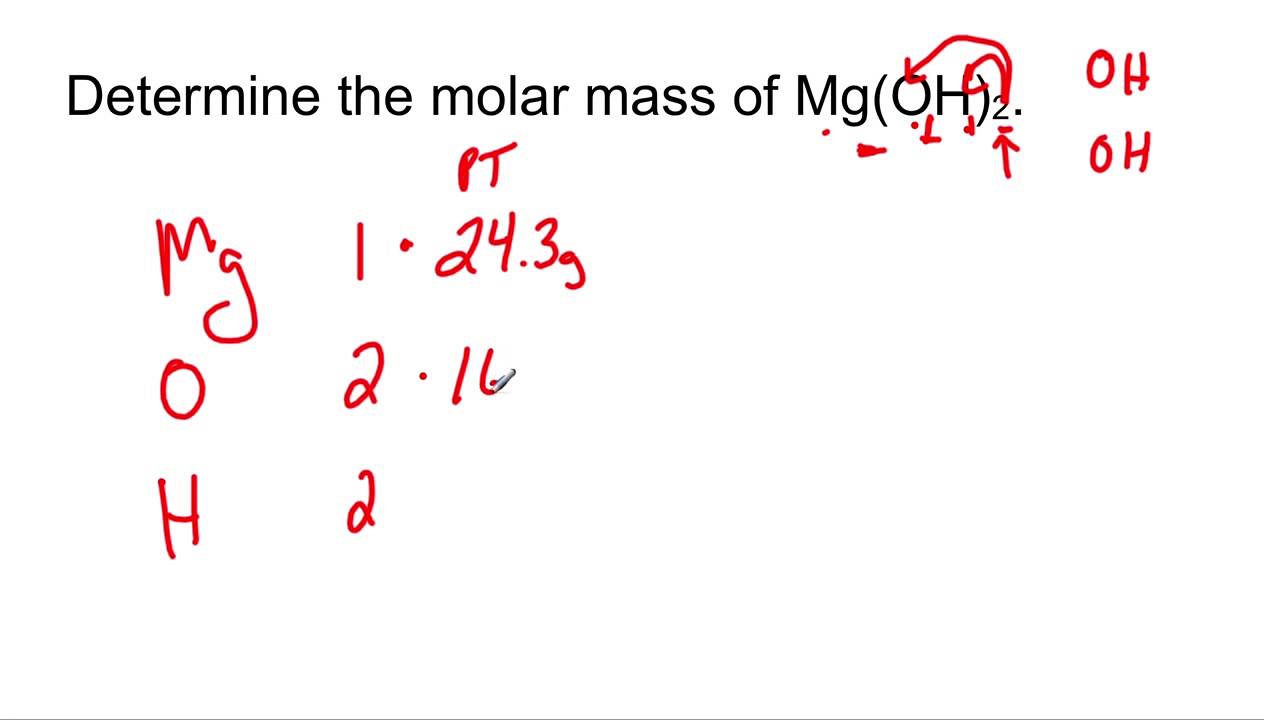SOLVED: The molar solubility of nickel(II) hydroxide (Ni(OH)2) is 2.8 x 10-6 mol/L in pure water at 25°C. What is the molar solubility of nickel(II) hydroxide in 0.050 M NaOH at 25°C? (
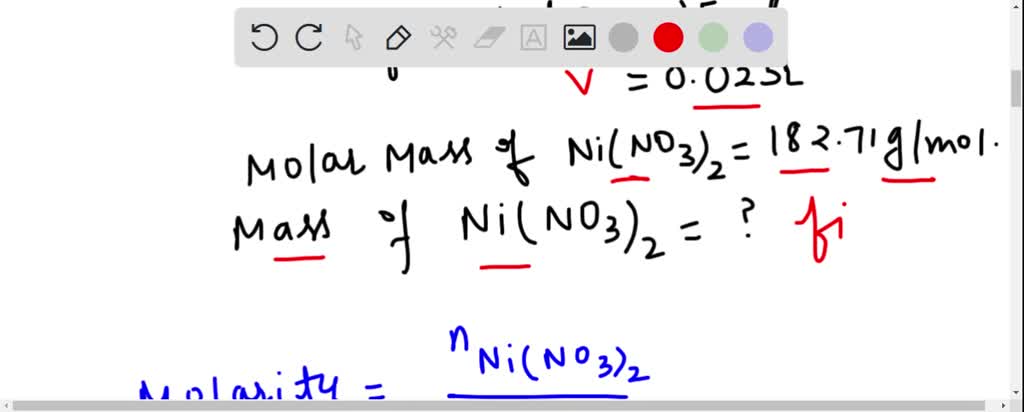
SOLVED: What is the mass of nickel(II) nitrate (182.71 g/mol) Ni(NO3)2 dissolved in 25.0 mL of 0.100 M solution? 4.00810.457 g 45.7 0.250 B 457 g
Unanswered Question 5You AnsweredCorrect Answer0/1 pts554. mL of 0.652 M KOH(aq) is mixed with 518. mL - brainly.com

MOLAR MASS The molar mass is the mass, in grams, of one mole (6.02 1023 particles) of an element (ion), a covalent molecule or a formula unit. molar. - ppt download

Pick Up a Packet & Calculator Write Down EQ Molar Mass EQ: How is molar mass calculated? - ppt download

Calculate the molar solubility of Ni(OH)2 in 0.10M NaOH. The ionic product of Ni(OH)2 is..... - YouTube

Ni(OH)2@Ni core-shell nanochains as low-cost high-rate performance electrode for energy storage applications | Scientific Reports
✓ Solved: Approximately 0.14 g of nickel(II) hydroxide, Ni(OH) 2(s) , dissolves per liter of water at...