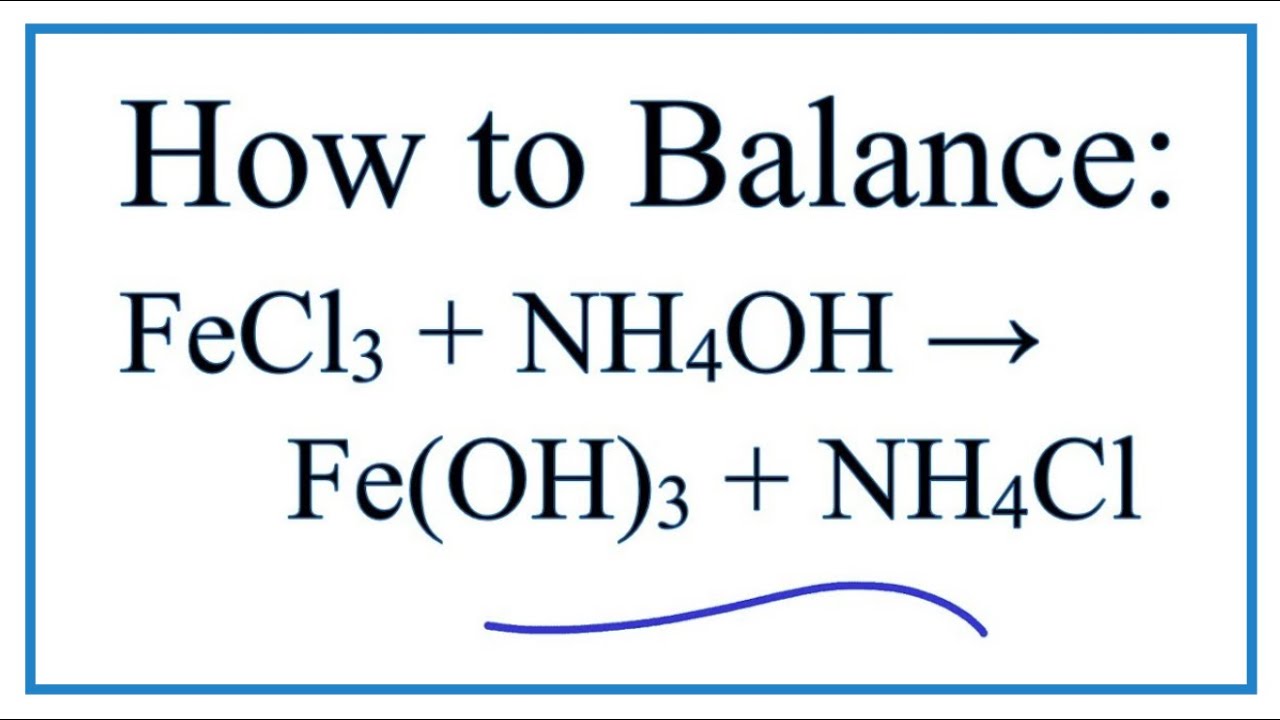
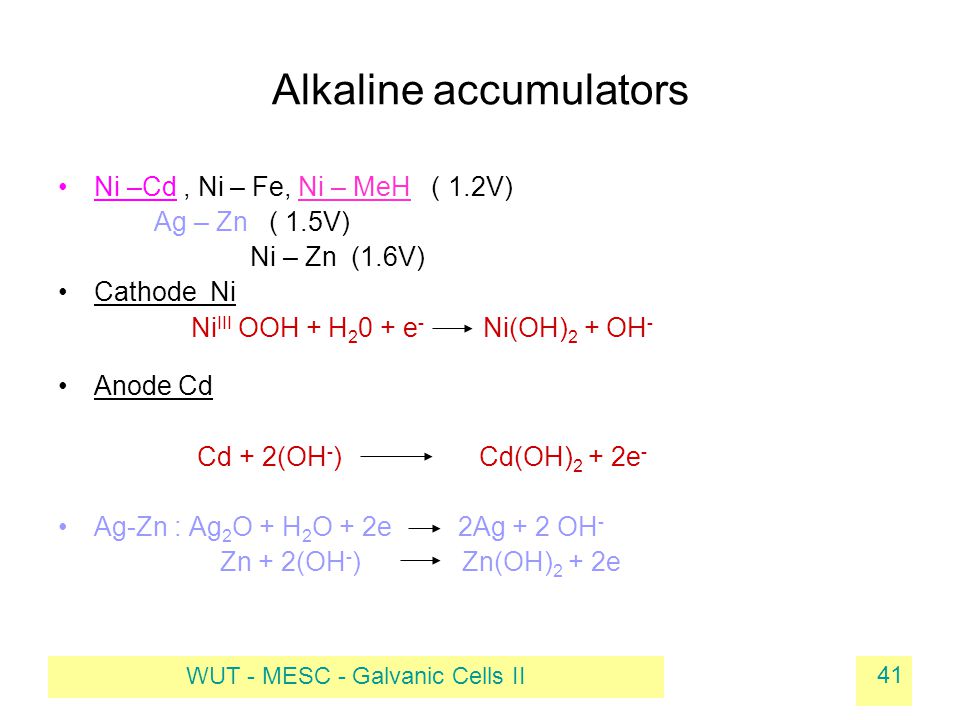
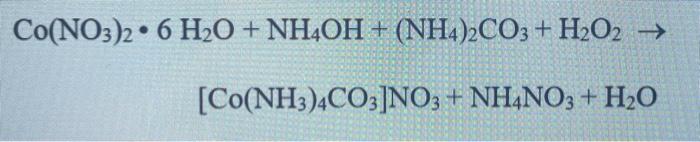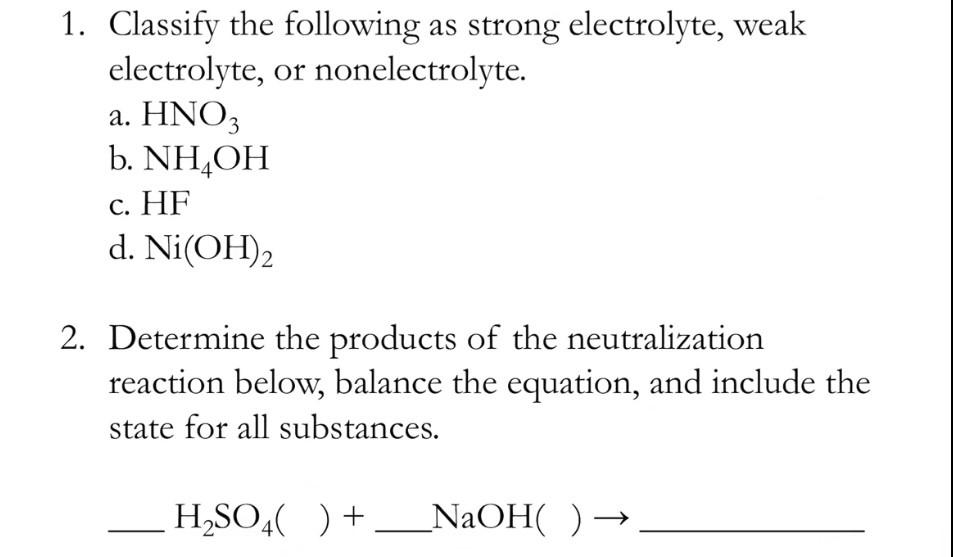SOLVED: When an aqueous solution of NH4OH is mixed with an aqueous solution of Ni(NO3)2, a pale yellow precipitate forms. Write a balanced molecular equation for this reaction. Write the complete ionic

TGA/DTA thermograms of Ni(OH) 2 prepared (a) without and (b) with CTAB. | Download Scientific Diagram

Figure 2 from Polyol-mediated synthesis of mesoporous α-Ni(OH)2 with enhanced supercapacitance. | Semantic Scholar

A 1 litre solution containing NH4Cl and NH4OH has hydroxide ion concentration of 10^-6 mol/litre. Which of the following hydroxides could be precipitated when the solution is added to 1 litre solution

Shape-controlled synthesis of Ni(OH)2/NiO nanowalls by surface reaction of Ni foil in aqueous NH4OH - ScienceDirect

Fabrication of Novel Hierarchical β-Ni(OH)2 and NiO Microspheres via an Easy Hydrothermal Process | The Journal of Physical Chemistry C

A 1 litre solution containing NH4Cl and NH4OH has hydroxide ion concentration of 10^-6 mol/litre. Which of the following hydroxides could be precipitated when the solution is added to 1 litre solution

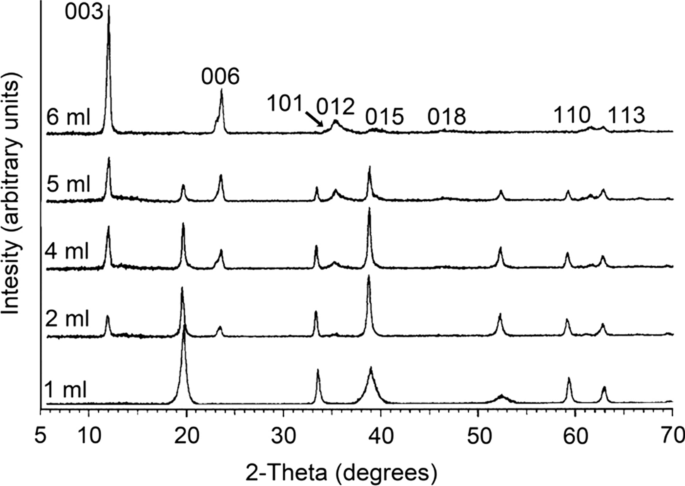
Direct preparation of Al-substituted α-Ni(OH)2 from Al-containing salt solution by immersing method - ScienceDirect
![PDF] Effect of reaction conditions on size and morphology of ultrasonically prepared Ni(OH)(2) powders. | Semantic Scholar PDF] Effect of reaction conditions on size and morphology of ultrasonically prepared Ni(OH)(2) powders. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/71d4ed12a62695a24100135a87df3664a7845d6e/2-Figure1-1.png)
PDF] Effect of reaction conditions on size and morphology of ultrasonically prepared Ni(OH)(2) powders. | Semantic Scholar